While the concept of providing extended safety data sheets through the supply chain is sound, the practice has been a failure.

This post is the first in a two-part series on chemical risk assessment. This one looks at the principles of risk assessment, particularly the chemical safety assessment required in the REACH Registration. We outline the process for the toxicological risk assessment, and its utility in providing users with risk management measures for using the chemical safely.
A second post will look at the use of chemical risk assessment within the workplace.
Risk assessment for fire and explosion is a separate topic.
If you wish to find out more about our services for risk assessment, see our services page or contact us. For help with the jargon, check our glossary.
What is chemical risk assessment?
Toxic chemicals can be safely managed if the exposure of humans and the environment is controlled.
The principle of risk assessment is that all chemical substances may be used safely, even if they are highly hazardous, provided that exposure of target organisms, notably humans, can be maintained below acceptable limits. This principle thus differentiates between risk, and hazard, as follows:
Risk = Hazard × Exposure (Equation 1)
This equation gives a useful qualitative appreciation of how risk can be managed through an understanding of hazards of a chemical (eg organ toxicity, skin irritation, toxicity to fish), and the numerous circumstances under which it contacts living things (exposure).
Toxic chemicals can be safely managed if the exposure of humans and the environment is controlled. The risk is not eliminated, unless there is no exposure at all.
The differentiation between hazard and risk has become controversial, with authorities under increasing pressure to ban or restrict use of highly hazardous substances, particularly carcinogens, mutagens, reproductive toxicants (collectively CMRs), endocrine disruptors, and other substances of very high concern (SVHCs) (see previous blog).
In general, such pressure comes from non-governmental organisations (NGOs), who argue that industry cannot be trusted to manage highly hazardous chemicals. Once chemicals are in the environment, it can be difficult to remove them, and they can disperse and enter food chains.
Indeed, some companies have caused widespread health and environmental problems through the release of hazardous chemicals. There have also been cases where the particular hazards of chemicals were not recognised at the time when the chemical was produced, with the problems being discovered only once the chemical was already widespread. A current example is very persistent, very mobile substances such as PFAS (see previous blog).
Industry favours using a risk-based approach to chemical safety to determine uses that cannot be shown to be safe, and so lead to restrictions of uses, rather than banning of the chemical because of its hazards. This allows chemical companies to continue using hazardous chemicals that might be difficult to substitute, provided that adequate measures are in place to protect workers and prevent release to the environment.
The current controls under the REACH Regulation [1] has a mixed approach. Manufacturers who register chemicals under the REACH Regulation at > 10 t/a must do a chemical safety assessment (CSA), ie a formal risk assessment with the results sent to the authorities. This identifies all uses throughout the chemical’s lifecycle, and the measures that should be in place to prevent harm. It can also identify uses that cannot be made safe.
But the REACH Regulation also bans or restricts substances are on the basis of their hazard. Substances identified as SVHCs are listed in Annex XIV of the REACH Regulation, and are banned from general use, and the authorities must give an authorisation for specific uses (see previous blog).
The REACH Regulation and Chemical Risk Assessment (CSA)
The REACH Regulation (either UK or EU versions) obliges registrants (usually the manufacturer, or the importer of a non-EU-manufactured substance; abbreviated as the M/I) supplying a substance at ≥ 10 t/year to supply a CSA to the authorities as part of the registration dossier (Article 14 of REACH).
The CSA contains summary information on the properties of the substance, obtained from testing, and other sources such as literature or structure–activity relationships.
Scheme 1 gives an overview of the CSA procedures for substances, from gathering information, to communication of the operating conditions, risk management measures, and the uses-advised-against to downstream users in the extended safety data sheet (ext-SDS). Further details of the process are given below.
The principles of CSA for substances also applies for chemical mixtures and releases from articles, but extra care is need to assess both the hazard (are there any synergistic effects?) and exposures (how is release of a substance from the mixture/article modified compared with the neat substance?). The reader is referred to the ECHA guidance in these circumstances. In the REACH Regulation, Article 14 and Annex I give the legal provisions for the CSA.
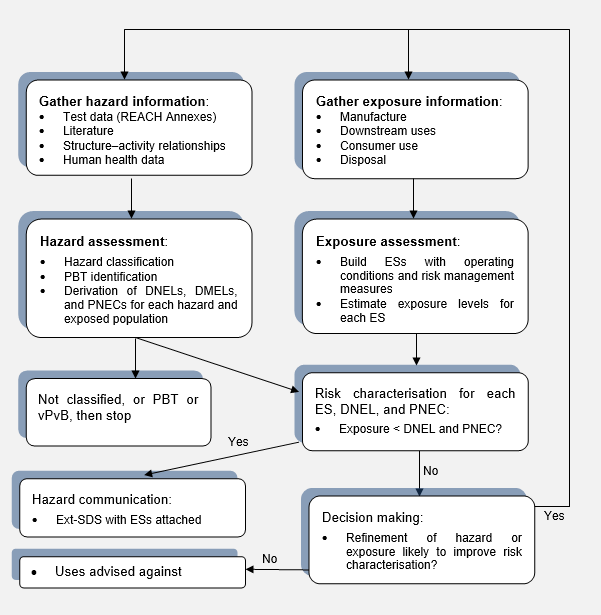
Hazard assessment
The hazard information is used to derive the classification of the substance according to the Classification, Labelling and Packaging Regulation (Regulation 1272/2008) [2] (see previous blog) and identify whether the substance is likely to be persistent, bioaccumulative and toxic (PBT), or very persistent and very bioaccumulative (vPvB), according to the criteria given in Annex XIII of REACH. If the substance is not classified as hazardous, and not identified as a PBT or vPvB, then there is no need to proceed further with the CSA.
For classified substances, or PBTs and vPvBs, the hazard data are used to derive ‘safe’ or ‘acceptable’ levels of exposure [derived no-effect levels (DNELs), derived minimal-effect Levels (DMELs) or predicted no-effect concentrations (PNECs)] for human or environmental exposure.
The more hazardous the substance is to health, the lower the DNEL (or DMEL).
The more hazardous the substance is to the aquatic environment, the lower the PNEC.
Exposure assessment
The manufacturer or importer (M/I) also has to gather information on the way that his substances are actually used in the supply chain, by checking with downstream users (DUs). Most chemical mixtures are developed with a particular use in mind, so understanding the use pattern may be straightforward, while for some common substances such as solvents there may be very many uses. Some DUs may regard their own or their customers use as confidential, and develop their own CSA (see later).
The use information is developed in Exposure Scenarios (ESs), which are activities (eg product transfer, mixing, cleaning) combined with the expected risk management measures (eg ventilation, enclosure, protective equipment) in order to determine potential human and environmental exposures. The lifecycle of the chemical will be considered during the assessment, eg:
- manufacture of the substance
- formulation into product
- industrial, professional, and consumer uses
- disposal
Each of the these steps may require activity ESs, eg for formulation:
- Transfer of substance to storage and reactors: hand pouring, automated pumping
- Mixing: hand mixing, reactor charging
- Sampling and analysis: hand sampling, laboratory use
- Packing
- Cleaning
The assessor may have to generate dozens of exposure scenarios.
The ESs are usually based on a use description [3] code system so that software, such as ECHA’s chemical safety assessment and reporting tool (CHESAR) can automatically quantify human and environmental exposures.
The calculations are based on default values for a reasonable worst-case scenario. If reliable measured values are available, these can be used to provide a refined exposure assessment.
The risk assessor then has the basic elements of Equation 1, ie the hazards of the chemical (converted to DNELs, DMELs and PNECs), and the exposures, and can therefore assess the risk to workers, consumers and the environment.
Risk characterisation
Risk characterisation is a comparison of the safe level (DNEL, PNEC) with the calculated exposure [4].
If the calculated or measured exposures are less than the DNELs, DMELs and PNECs, then the use is safe.
If the exposure are higher, then the use is unsafe. The assessor then has the choice of prohibiting the use, or of re-calculating the risk characterisation by:
- Refining hazard data, eg by commissioning more testing to reduce uncertainties, with the expectation of raising the DNEL, DMEL, or PNEC
- Refining exposure data, eg by measurement, or stricter control measures or changing the operating conditions.
Communication in the supply chain
The M/I, having arrived at ESs (ie conditions which allow safe use of the substance for each of the foreseen activities), has to communicate these to the downstream users (DUs). The main tool of communication is the SDS. The ESs relevant to the DU are attached to the SDS as an Annex, giving a so-called extended safety data sheet (ext-SDS).
The problem with is that the ESs have proven to be of limited utility.
Other key parameters are given in Section 8 of the SDS (Exposure controls/personal protection), particularly the risk management measures (eg PPE, ventilation, and environmental controls). This Section also contains the DNELs, DMELs and PNECs, along with occupational exposure limits, giving the upper limits of exposure for inhalation, skin, and sometimes ingestion.
Section 1.2 of the SDS (relevant identified uses of the substance or mixture and uses advised against) states the ‘uses-advised-against’, ie uses that cannot be demonstrated to be safe in the CSA.
The data sections of the SDS, physico-chemical (Section 9), toxicology (Section 11) and ecology (Section 12), should be consistent with the data in the CSA.
While the concept of providing extended safety data sheets through the supply chain is sound, the practice has been a failure. The problem with is that the ESs have proven to be of limited utility. The ES attached as an Annex are often machine written with a lot of jargon, and use a non-intuitive coding to describe simple uses. For common substances, the ext-SDS can have a dozens of ESs and the Annex can run to hundreds of pages. When supplied with such a document, the downstream user often lacks the time or expertise to understand and implement the measures, which the supplier has so carefully determined in the CSA.
Downstream user considerations
If the ext-SDS is understood, the DU will be able to determine the risk measurement measures necessary for safe use of the substance. As indicated previously, the DU should comply with these conditions of use, or ask the M/I to modify the ES, or conduct their own CSA to determine their own ESs for their use.
The DU, aware of their own customers confidential use, may also wish to conduct a CSA for these uses. CSAs covering new uses should be submitted to ECHA.
A formulator may receive substance ESs from each ingredient supplier. The formulator can add ingredient ESs to his mixture SDS. Alternatively, the formulator may help his customers to implement proper risk measurement measures by combining the ingredient ESs into a mixture ES, or by incorporating the information into the body text of the SDS.
The principles involved in developing a CSA for mixtures in this case are the same as those outlined above. The DU will need to take into account hazards posed by the combination of ingredients, such as changes in physical state or synergistic or antagonistic effects.
References
[Back to What is risk assessment] [Back to Hazard assessment] [Back to Exposure assessment] [back to Risk characterisation]
[1] Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) (as amended).
[2] Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures (as amended).
[3] Guidance on Information Requirements and Chemical Safety Assessment; Chapter R.12: Use Description; ECHA, December 2015.
[4] Guidance on Information Requirements and Chemical Safety Assessment, Part E: Risk Characterisation; Draft (Public) Version 3.0; March 2016.
2 comments
Comments are closed.